Hepatitis A: A Clinical Spectrum of the Disease in Children Admitted in a Tertiary Care Hospital
Original Research
Corresponding author: Jeetendra Gavhane, Professor, Department of Pediatrics, MGM Hospital
and Medical College, Kamothe, Navi Mumbai, Maharashtra, India.
Email: drjeetendra@gmail.com
Telephone no: 9870106094
ABSTRACT
Introduction: Hepatitis A virus (HAV) is a common communicable disease acquired from feco-oral route. It is endemic in fast developing urbanization areas. Poor sanitation is making this disease stay despite good vaccine availability. It may present with a range of clinical manifestations, from asymptomatic infection to fulminant hepatitis.
Aims: To study the clinical profile of children with Hepatitis A infection.
Materials and Methods: It’s a retrospective analysis of children having Acute HAV diagnosed by IgM hep A in 6 months to 12 years of age in last one year in tertiary care hospital. Clinical features, complications were studied. Results: 27 cases were enrolled in our study. Mean age was 6.5 ± 3.9 years out of 27 cases. Fever (92.6%), gastrointestinal complaints (59.3%) and lethargy (37%) were the common presenting symptoms. 12 patients (44.4%) presented with irritability and altered sensorium. Physical findings were hepatomegaly (96.2%) and jaundice (100%) in all children. More than 4-fold increase in level of Aspartate transaminase and Alanine transaminase were noted in almost all cases. Prothrombin time was deranged in 12(44.4%) cases. Serum Ammonia was raised in 8(27%) patients. 8 cases (27%) developed fulminant hepatic failure out of which 2 cases had co-infection with dengue, 1 case had a history of consumption of nimesulide and 1 case had chronic liver disease & 7 cases (25.9%) succumbed to death. Rest 20 (74.1%) recovered completely. Conclusion: Though hepatitis A infection is a self-limiting disease, presence of co-infection, underlying liver disease and consumption of certain drugs can cause severe complications. Hence, all cases should be followed up till complete recovery.
Keywords: Hepatitis A virus.
Introduction
In India, Acute Hepatitis A (HAV) has been identified as the most common etiological agent for sporadic Acute Viral Hepatitis, especially in the pediatric age group.1 However, no age is immune to this virus. It is a common viral infection and is spread is principally via the faeco-oral route and due to poor hygiene and sanitation is still endemic in many developing countries. The clinical spectrum of acute hepatitis A infection is varied. It can either be asymptomatic or present with a range of clinical manifestations, which may include atypical presentations like relapsing hepatitis, cholestatic hepatitis, and extra hepatic manifestations in children. Over the last few years, number of cases with atypical presentation of hepatitis A in children has also been increasing. Here we present a study aimed to analyze the clinical features, laboratory parameters and other probable prognostic factors of HAV of pediatric population attending a teaching hospital in Navi Mumbai.
Method
A cross-sectional study was conducted in Department of Pediatrics, Mahatma Gandhi Mission Hospital from December 2018 and November 2019. Patient from age group 0 to 15 years presenting with signs and symptoms of acute viral hepatitis such as loss of appetite, jaundice, nausea, vomiting, pain abdomen and itching were investigated for HAV. Patient with positive HAV IgM were taken in our study. Patients were admitted for observation and further management. The indications for admission were poor general condition, not being able to eat properly due to protracted vomiting, high fever and altered sensorium. Patient managed on OPD basis were excluded from our study. The consent for the study was taken from parents of the children. Patients were then subjected to blood tests like complete blood count, liver function tests, prothrombin time and serum ammonia in cases with altered sensorium. Cases were managed conservatively with symptomatic treatment and dietary advice. Antibiotics were used whenever required. Statistical analysis and data record were done using Microsoft Excel 2010.
Results
A total of 27 patients under the age of 15 years attended the department of paediatrics in MGM hospital with acute hepatitis A. The average age of patients was 6 years in our study (1-12 years) and most cases were in 0-5 years age group. Male patients were predominant. Most them had history of consumption of outside food. None of them were immunized with hepatitis A vaccine. One child had a history of consumption of nimesulide and two children also had dengue as co-infection. Demographics according to various age groups from this data can be seen in Table 1.
Signs and Symptoms – The presenting complaints of the studied subjects were variable and are given in Graph 1. The most common presenting complaints were jaundice (100%), fever (92.6%), dark coloured urine (81.4%) gastrointestinal complaints like diarrhoea (55.5%), vomiting (22.2%) and pain in abdomen (59.3%) and lethargy (37%). 12 patients (44.4%) presented with irritability and altered sensorium. There was overlap of many symptoms. Most common physical finding was hepatomegaly (96.2%). One patient (3.7%) developed ascites. Eight patients (29.6%) suffered from fulminant hepatic failure. All were treated by conservative management in ICU set up. Seven patients (25.9%) unfortunately succumbed to death out of which 2 cases (7.4%) had co-infection with dengue, 1 case (3.7%) had a history of consumption of Nimesulide and 1 case (3.7%) had chronic liver disease. There were no re-admissions.
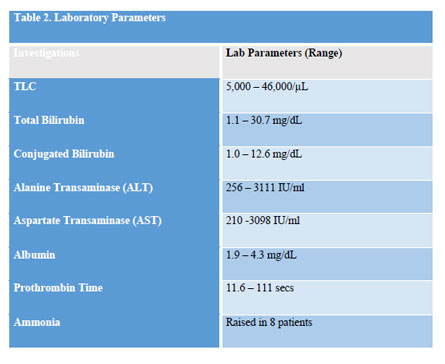
Laboratory test – Along with the confirmation HAV IgM test patient’s other investigations were also sent such as CBC, liver function test, prothrombin time and levels of serum of ammonia were also checked in patients with altered sensorium. The values of the various laboratory investigations were variable. 16 cases (66.6%) showed leucocytosis (TLC > 10,000/μL). Total serum bilirubin and direct bilirubin was raised in all children (Serum Bilirubin >1 mg/dL). More than 4 fold increase was seen in Alanine Transaminase (ALT) and Aspartate transaminase (AST) was seen in almost all cases (92.5%). Low serum albumin was noted in 2 cases (7.4%). 12 cases (44.4%) had deranged prothombin time of more than 15 seconds. Serum ammonia levels were done in patient who presented altered sensorium. 8 patients (29.6%) showed raised levels (normal 30 – 86 mcg/dL). Laboratory parameters are given in Table 2.
Recovery – From the 27 patients, 21 cases (77.7%) showed complete recovery. 8 cases (29.6%) had developed acute fulminant hepatic failure and unfortunately 7 cases (25.9%) succumbed to death. Only supportive treatment (diet, bed rest) was recommended during the course of the acute illness. Patient who had developed acute fulminant hepatic failure were treated in ICU care.
Discussion
In India, HAV continues to be endemic and the most common type of acute viral hepatitis where it was found that out of 172 studied, 111 (64.5%) of children were found to have suffered from HAV.1 The symptoms and signs of acute hepatitis are varied and are not very diagnostic in distinguishing between the different hepatitis viruses. Hepatitis A itself presents as completely asymptomatic to fulminant hepatic failure in the paediatric age group. In our 27 hepatitis A patients, fever was the most common presenting symptoms in 92.6% and diarrhea in 56% of cases which can give a hint to the paediatrician in the diagnosis of a child with HAV. This finding was similar to few studies which showed fever and diarrhea to be the presenting complaints.2-4 Many children initially presented with low grade and had attended on OPD basis for acute febrile illness but eventually developed jaundice hence slightly delaying in the diagnosis of acute hepatitis A. Jaundice (100%) and hepatomegaly (78%)were the common physical findings. Ascites was seen in 1 patient (3.7%) which could be due to the presence of low serum albumin. A study done in India reported ascites to be an atypical finding.5 The mean presenting laboratory findings such as bilirubin, AST, and ALT levels in our hepatitis A patients were similar to those done in the other studies.2-4 Patients with high bilirubin levels, had a longer course of illness and it took a longer time for the bilirubin to come back to normal levels. The raised level of AST and ALT did not determine the severity of the cases. On an average it took almost 2 weeks for the patient’s biochemical profile to reach it normal levels with one patient taking almost 8 weeks to completely recover. There were no relapses of the infection in our study but the children did frequently complain of abdominal pain during the recovery phase. Generally acute Hepatitis A infection has a very favorable outcome but unfortunately in our study the incidence of children developing acute fulminant liver failure was very high. It was seen in 8 cases i.e. 29.6% of patients. Even mortality rate of the patients were high as 7 children (25.9%) did not make it. This number is quite high compared to other recent study from India where the mortality was just was 1.3%.6 Such results could be due to a short span of time of study with a small sample size which was a limitation of our study. Also the children who presented with fulminant hepatic failure had history of seeking late medical attention, one had history consumption of banned drug like nimesulide, two cases had presence of co-infection with dengue and one patient being a case of chronic liver disease who had presented to us an relapse of the HAV suggesting these could be the risk factors for children with acute hepatitis A.
Conclusion
Though, Hepatitis A is is a self-limiting disease but presence of certain risk factors such as faulty dietary habits, consumption of certain drugs, co-infection and underlying liver disease can cause severe complications. Hence, all cases should be followed up till complete recovery.
Funding: No Funding Sources
Conflict of Interest: None declared
Ethical approval: The study was approved by institutional ethics committee.
References
1. Thapa BR, Singh K, Singh V, Broor S, Singh V, Nain CK. Pattern of hepatitis A and hepatitis B virus markers in cases of acute sporadic hepatitis and in healthy school children from North West India. J Trop Paediatr. 1995;41:328-9.
2. Koff’ RS. Clinical manifestations and diagnosis of hepatitis A virus infection. Vaccine 1992; 10(suppl 1 ):S 15-7.
3. Ciust ID. Clinical features. In: Gust ID. Feinstone SM. eds. Hepatitis A. Boca Raton. FL: CRC Press. 1988:145-62.
4. Seeff LB. Diagnosis, therapy, and prognosis of viral hepatitis. In: Zakim D. Boyer TD. eds. Hepatology: a textbook of liver disease. 2nd ed. Philadelphia: WB Saunders. 1990:958-1025.
5. Kamath SR, Sathiyasekaran M, Raja TE, Sudha L.Profile of viral hepatitis A in Chennai. Indian Pediatr. 2009 Jul;46(7):642-3.
6. Kumar A, Yachha SK, Poddar U, Singh U, Aggarwal R. Does co-infection with multiple viruses adversely influence the course and outcome of sporadic acute viral hepatitis in children? J GastroenterolHepatol. 2006 Oct;21(10):1533-7.
Issue: July-September 2019 [Volume 8.3]